FDA Tells At-Home Diagnostics Companies To Stop Coronavirus Test Roll-Outs
The companies are complying. Customers won't get their results and are being told to destroy their test kits.
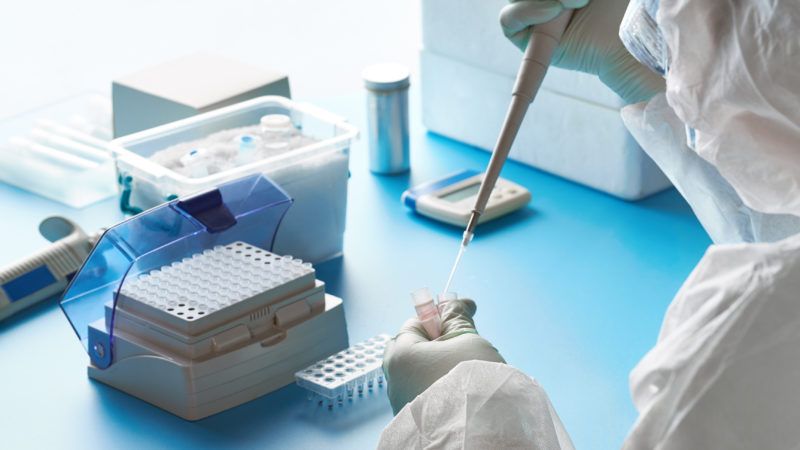
Four different diagnostics companies were planning to ship at-home tests for the coronavirus infections to their customers this week, but are instead "pausing" their roll-outs in order to avoid the displeasure of Food and Drug Administration (FDA) bureaucrats. In fact, companies are destroying samples sent back from customers rather than testing them to comply with FDA guidance.
The companies had launched their tests based on their interpretation of the latest FDA Emergency Use Authorization (EUA), which says that any lab that had been certified under the appropriate quality-control standards could start performing coronavirus tests. They were all using just such labs.
For example, to comply with the FDA's testing guidance TechCrunch reports that the California-based Carbon Health sent out a notice to its coronavirus at-home test customers stating that "we sincerely regret to inform you that you will not get a test result. If you have already shipped your kit back, the specimen will be destroyed by Curative, Inc [their lab testing partner] using standard biohazard disposal. If you have not received your kit yet, please discard it upon receipt."
Austin-based Everlywell was ramping up to handle 250,000 of its at-home coronavirus tests per week. Since the FDA's EUA does not now stop private labs from developing and deploying coronavirus tests for clinical use, the company has pivoted to making its initial supply of COVID-19 tests available to hospital and health care providers, as a way to get around the FDA's regulatory roadblock. Carbon Health is offering their coronavirus testing at its walk-in clinics.
The fact that self-administering these tests is uncomfortable may make their results less accurate, but the fact that the FDA allows their use by health care providers indicates that these tests work and are in no way fraudulent. The FDA needs to get out of the way of at-home COVID-19 testing now.

