A 'Right to Try' Bill Is Finally Heading to Trump's Desk
It's still not clear whether pharmaceutical companies will work with patients outside the FDA's supervision.
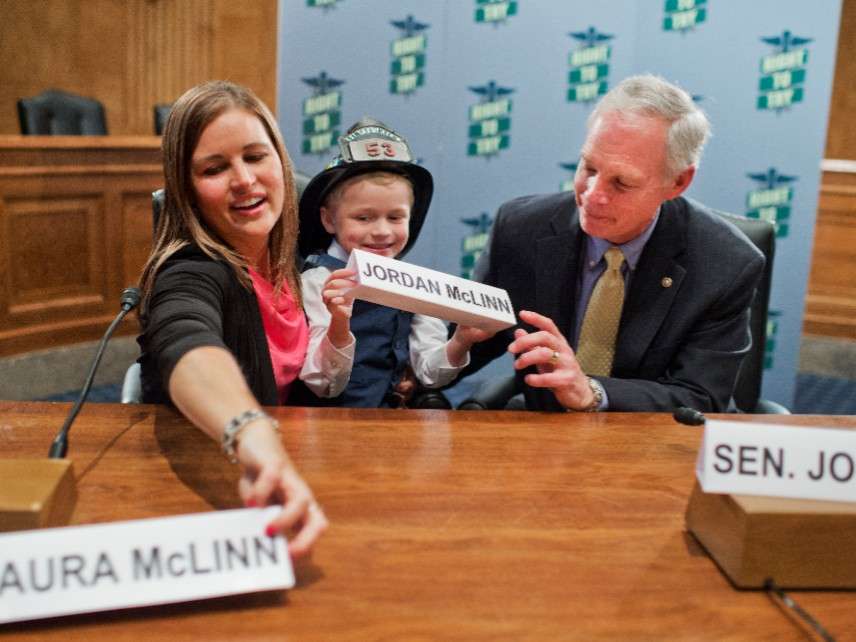
After months of debate, the House of Representatives on Tuesday passed a bill that will allow patients with life-threatening diseases to use experimental treatments without the approval of the Food and Drug Administration. The Trickett Wendler, Frank Mongiello, Jordan McLinn, and Matthew Bellina Right to Try Act of 2017, which the Senate approved in August 2017, now heads to President Trump, who has promised to sign it.
To be covered by the bill, treatments have to complete Phase 1 of the clinical trial process, which is the most basic safety assessment. The bill does not compel doctors to participate in Right to Try treatment protocols. Nor does it require pharmaceutical companies to provide experimental treatments to patients who request them.
An earlier House version of the bill was restricted to patients who had reached "a stage of a disease or condition in which there is reasonable likelihood that death will occur within a matter of months," or who were suffering from a disease "that would result in significant irreversible morbidity that is likely to lead to severely premature death." The version approved this week, by contrast, covers anyone with a "life-threatening disease."
The bill provides some legal protection for both doctors and drug companies in the event that an experimental treatment hastens a patient's death or causes some other undesirable side effect. The bill also requires drug companies to report any adverse effects to the FDA. Right to Try proponents say reporting such outcomes will not affect the approval process for the drug unless an adverse reaction reveals a fundamental danger.
Whether passage of this bill is a good news for patients depends on whom you ask. Medscape has rounded up reactions from various corners of the health care world. The conventional wisdom says no one should consume anything that has not completed all three phases of FDA clinical trials and been approved by the agency. A sample reaction:
The legislation "opens the gate to a dangerous, uncharted pathway for accessing experimental medications that have not been shown to be safe or effective," said Michael A. Carome, MD, director of the Health Research Group at Public Citizen, in a statement. "The bill passed today will expose vulnerable patients to risks of serious harm, including dying earlier and more painfully than they otherwise would have, without appropriate safeguards."
Carome and other opponents of the bill argue that the FDA's "expanded access" program (a.k.a. "compassionate use") does everything Right to Try would while providing an additional layer of patient protection. Other critics have argued that Right to Try will divert patients from clinical trials. These talking points are at odds with each other: If patients don't need Right to Try because they can already obtain unapproved treatments through expanded access, but expanded access is not draining the pool of clinical trial participants, why would Right to Try?
The libertarian Goldwater Institute, a leader in the contemporary Right to Try movement, is very pleased:
"Today's vote is a win for patients. Millions of Americans who have been told they are out of options and it's time to get their affairs in order, are closer to having the opportunity for one last treatment, without having to get permission from the federal government first," said Victor Riches, president & CEO of the Goldwater Institute. "Members of Congress came together to put patients first and we're grateful for their support for this bipartisan, grassroots movement powered by real patients in all 50 states."
The free market Heartland Institute also supports the bill. "Now, at long last, American patients and their families can have hope that a life-saving drug will no longer be denied to them because of bureaucratic barriers," Heartland President Tim Huelskamp said in a statement emailed to reporters. "They will no longer be barred from trying to save their lives."
I think Robert Graboyes at the Mercatus Center has the most concise summary, contained in an email from Mercatus, of what's at stake:
Effectively, the law grants patients, doctors, and drug manufacturers greater decision-making authority and greater capacity to assume risks. The practical importance of the law remains to be seen. Opponents of the new law argue that the FDA's compassionate use waivers already get investigational drugs into patients' hands early on; supporters argue that the administrative burdens of the waiver program discourage its use. Now, we'll get a better idea of who is correct.
Results will be interesting. The FDA has always had a seen-and-unseen problem in its incentives. Patients who suffer adverse outcomes from using experimental drugs are relatively easy to identify, while those who suffer for lack of experimental drugs are largely unidentified and unseen. In more concrete terms, it's easier to point a TV camera at the first group than it is at the second group. That asymmetry could still frustrate the intent of the new law. Time and experience will tell.
Earlier this year, I dug a little deeper into why a federal Right to Try bill might not move the needle much. The short version is that incentives are aligned for pretty much everyone except the drug companies. Some patients might get to try treatments that could extend their lives, and their doctors would get to bypass insurance companies (because no insurer is going to touch a Right to Try treatment) and forgo the FDA's paperwork.
But I'm not sure what's in it for drug companies. Right to Try data won't be randomly controlled, which means they won't help make the case for FDA approval. An adverse outcome, meanwhile, could hurt a drug's chances of approval even if its role in that outcome is hard to measure in an uncontrolled treatment setting. As Graboyes says, the only thing we can do now is wait for the market to react and hope for the best.

