FDA's Vaping Regulations Will Hurt Smokers Trying to Quit
Because lawmakers didn't understand that the future might bring new, better products, we'll soon be stuck with only the old, dirty options.

Electronic cigarettes are now the most popular technique used by Americans who want to quit smoking. But that pathway could close later this year, thanks to shortsighted federal regulations that effectively prevent innovation.
When Congress passed the Tobacco Control Act in 2009, few electronic cigarettes were on the market. Under the terms of that law, the Food and Drug Administration (FDA) would have the authority to approve or deny any new tobacco products introduced after February 15, 2007, while products that had been on the market before that so-called "predicate date" would be free from the new level of scrutiny.
That works out fine for cigarettes, cigars, chewing tobacco, and other items that have been around a long time, but it effectively froze the market. Any new products—including almost all vaping devices and the nicotine-laced liquids used in those devices—would have to go through an expensive and vague regulatory process before being offered to consumers.
The deadline for filing those applications is November 8 of this year, unless Congress and the FDA act to change the rules and let e-cigarettes remain on the market.
Greg Conley, president of the American Vaping Association, explains it like this. If you have not filed a retroactive application for any vapor product that has come to market since 2007—which is every single product on the market today—your product is banned. If you file an application before November 8, and the FDA doesn't like what you have included, you're banned. If you file an application on November 8, and the FDA hasn't ruled on that application by November 8, 2019, you are banned.
"So you could spend millions and millions of dollars to try to comply with very vague requirements that have been put out by the FDA, and the FDA could still simply never review your application or just turn it down for an arbitrary reason," Conley said at a recent forum sponsored by the American Enterprise Institute. The FDA's own economic analysis of the regulation suggests that 98 percent of all e-cigarette products will not apply to stay on the market.
That's bad news for vaping businesses, but it's also bad news for Americans hoping to stop smoking cigarettes.
According to research from the Center for Disease Control, 35 percent of Americans who sought to quit smoking from 2014-2016 used electronic cigarettes as a substitute. Vaping allows would-be smokers to get a hit of nicotine and to maintain the same physical routine, while avoiding the dangerous chemicals and soot that come from burning tobacco and inhaling it into their lungs. Compared to other methods used to quit smoking, the CDC reports, e-cigarettes are the most popular, beating out nicotine gum, anti-smoking patches, and FDA-approved medications such as Zyban and Chantix:
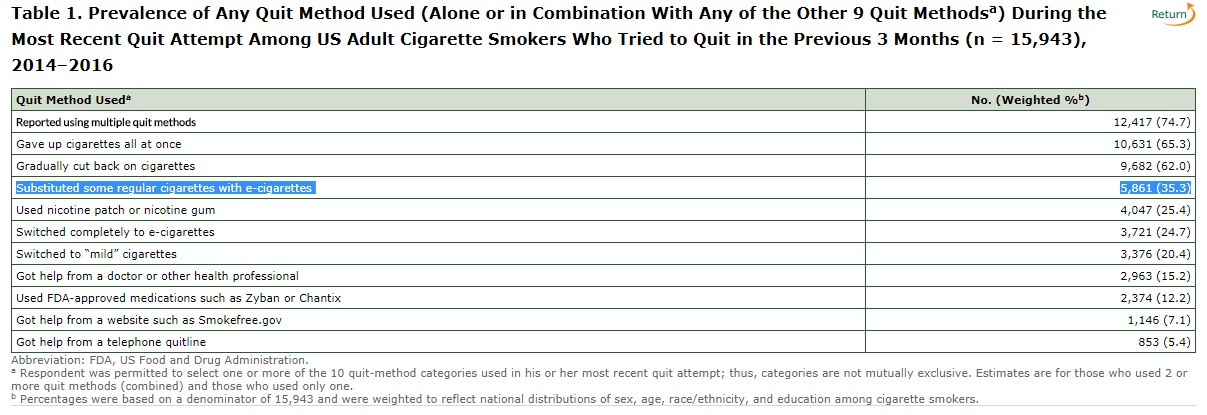
Killing the majority of vaping products currently available on the market while leaving cigarettes available is almost certain to drive some e-cigarette users back to combustible tobacco options. That means the FDA—the very government agency that claims it is "responsible for protecting the public health" and "for advancing the public health by helping to speed innovations"—will be banning innovative products that are helping Americans improve their health. They'll be doing that because Congress, a decade ago, made an arbitrary decision that tobacco products made after 2007 should have to face a different level of scrutiny than those that came earlier.
Imagine how a similar rule would effect any other industry. What would computers look like today if Congress decided in 1999 to force any new microchip-using devices to jump through additional regulatory hurdles while leaving older models on the market? What car would you be driving today if any innovations to internal combustion engines were banned in the 1970s?
It's the same story with electronic cigarettes. Because lawmakers lacked the foresight to understand that the future might bring new, better products, Americans might soon be stuck with only the old, dirty options.

