FTC Thinks Emphasizing That Four Loko Is 5 Drinks in One Can Will Encourage Moderation
This week the Federal Trade Commission (FTC) issued a final order compelling Phusion Projects, the manufacturer of the fruity malt beverage Four Loko, to change its packaging so that consumers will finally know how totally messed up you can get by polishing off an entire 23.5-ounce can. Was there ever any real confusion on this score? Since volume and alcohol content were clearly listed on every can, and since the FTC complained that Phusion Projects blatantly promoted Four Loko as a fast and economical way to get drunk, probably not.
Yet somehow the FTC became convinced that Phusion Projects "represented, expressly or by implication, that a 23.5 oz can of 11% or12% ABV Four Loko contains alcohol equivalent to one or two regular, 12 oz beers." It must have been a pretty subtle implication, because if you look through the FTC's original complaint you will see that the company never said anything to that effect. "We do not agree with the FTC's allegations regarding these issues," Phusion co-founder Jaisen Freeman said in a statement, but "we consider this agreement a practical way to move forward." The main allegation against Freeman's company boils down to this: By putting 23.5 ounces in one "nonresealable" container, Phusion Projects implied that it was OK for one person to consume it all in one sitting, when public health experts tell us you really shouldn't do that.
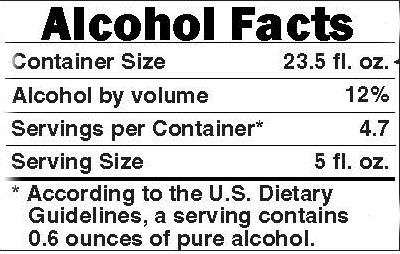
A can of Four Loko contains less alcohol than a bottle of Champagne and less alcohol than some big bottles of craft beer. In terms of intoxicating power, it is equivalent to a few cocktails—or, as the FTC prefers to put it, "4.7 regular beers," by which it means beers with an alcohol content of 5 percent. That is an amount people have been known to consume over the course of an evening without any deleterious effects. But let us accept the FTC's premise that no one should be drinking a whole can of Four Loko by himself. How do the packaging changes it has foisted upon Phusion Projects serve to discourage that allegedly reckless practice? First, Four Loko containers henceforth will be resealable, which should be quite effective if the desire to avoid waste, as opposed to the desire to get wasted, is the main reason people drink the entire can. Second, Four Loko containers will carry an "alcohol facts" panel listing, in addition to the volume and strength information that was on the old cans, "servings per container" (4.7) and serving size (five ounces), accompanied by this statement: "According to the U.S. Dietary Guidelines, a serving contains 0.6 ounces of pure alcohol."
Assuming that Four Loko consumers care how many ounces of pure alcohol the federal government deems to be a serving, and assuming they are prepared to do the math required to convert pure alcohol ounces into beverage ounces based on alcoholic strength, that last piece of information will be very useful to them. Likewise "serving size," assuming they are pouring their malt beverage into a measuring cup. As for the declaration that one can is 4.7 servings, one can imagine how it might backfire. As I suggested in Reason last year, advertising that a can of Four Loko is equivalent to five drinks calls further attention to the product's most controversial attraction.
Apparently I was not the only person to think of that. The FTC originally proposed a more prominent statement saying, "This can has as much alcohol as 4.5 regular (12 oz. 5% alc/vol) beers." But Reuters reports that the commission thought better of that idea:
The FTC said it had earlier considered requiring alcohol information on the fronts of cans. But it said some commentators feared this could encourage binge drinking, by promoting Four Loko as "an efficient, inexpensive way to become inebriated."
Solution: Put the same information on the can, but make it so inconspicuous that no one will notice it. This is the FTC's idea of a balanced approach to consumer protection.
The FTC's order is the second federal crackdown on Four Loko. The first, by the Food and Drug Administration, resulted in its decaffeination, through a completely rational, science-based process that I chronicled in Reason two years ago.
