Americans Get Paid To Donate Plasma. Everyone Else Should Too
Our secret weapon against COVID-19 could be cold, hard cash.
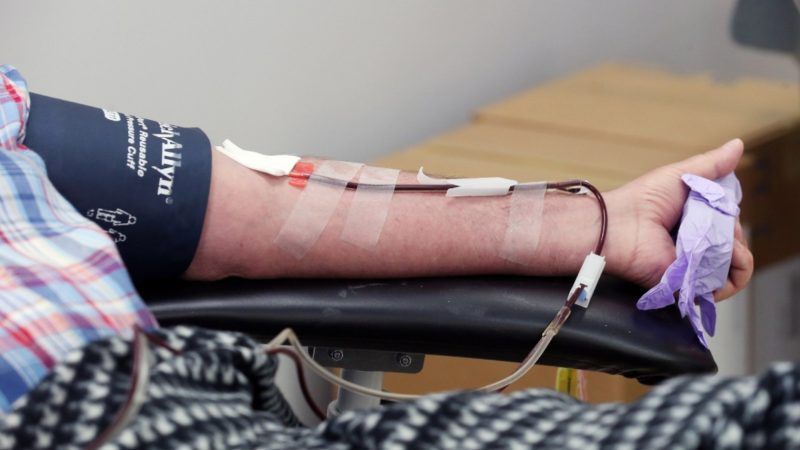
Life-saving plasma therapies are essential for many patients, but every year we flirt with a shortage. Plasma collected in the United States is the source material for more than 70 percent of the global supply; humanity is nearly always one disruption in the U.S. plasma supply away from global catastrophe. This constant near-crisis is a result of most foreign countries' refusal to pay people for plasma used to manufacture therapies—the consequence of foolish adherence to decades-old, outmoded guidance from the World Health Organization (WHO).
You may have already heard that blood donations are down significantly because of the coronavirus pandemic. So are blood plasma donations, which may actually prove to be the bigger problem in the long run. Plasma donations have fallen 15–20 percent across the United States. Texas and Florida, the two states with the most plasma centers, are once again seeing an uptick in coronavirus cases, threatening to further depress donations at those states' plasma centers.
This is a threat to the thousands of patients not just in the United States, but also to tens of thousands of patients around the world who are dependent on American plasma. Blood plasma is used to make plasma therapies—or plasma protein therapies—including immune globulin (or Ig), albumin, C-1 inhibitor, and clotting factor, among others. These therapies are then used to treat people with immune deficiencies and rare blood disorders, like Von Willebrand disease and hemophilia. For many, these are life-saving treatments that they cannot go without.
Plasma is a key ingredient in a possible therapy for COVID-19 itself. The plasma industry is working on a promising hyperimmune globulin treatment for the coronavirus. Transfusions of convalescent plasma are undergoing trials in many countries with plenty of early successes being reported. Should this work, it will likely be due to the introduction of antibodies from those who have recovered from COVID-19 into those whose immune system is still trying to figure out how to make antibodies to fight off the infection. This is a shotgun approach—the patient receives all of the components and proteins present in convalescent plasma, and not just the antibodies. A hyperimmune globulin is a laser—it delivers the isolated and concentrated antibody and nothing else.
Between the depressed supply of plasma and the possible future uptick in demand, the world could well be heading for a devastating shortfall. The problem could be quickly and cheaply remedied if it weren't for irresponsible guidance from global health bodies and an unfounded bias against paid donation.
Our Precious Bodily Fluids
American plasma is the lifeblood of this $26 billion global industry. Plasma represents 1.6 percent of American's total exports and nearly three-quarters of the total global supply. With demand for these therapies growing at 6-10 percent each year for more than a decade, it has risen to become America's 11th-largest export category. Demand is projected to keep growing at those rates for the next decade and beyond, even before you factor in the impact of COVID-19. Five percent of the world's population provides more than half of the world's plasma.
American dominance in the plasma market is explained by one simple fact: In America, it is legal and commonplace to pay people to give plasma. Millions of Americans regularly give plasma in exchange for $30 to $50 per donation. The average American donor gives 21.4 times per year, with a per capita collection volume of 113 liters of plasma per 1,000 people. If you add plasma obtained from Germany, Austria, Hungary, and Czechia—the other places where a form of compensation (typically capped at 25 euros, intended only to cover expenses) is offered—paid plasma accounts for a staggering 89 percent of all the plasma used to make plasma therapies for the whole world. Just five countries account for nine-tenths of the world's plasma.
Thanks to the four European countries that pay for plasma, Europe is only about 40 percent dependent on the U.S. at the moment. But that figure is growing quickly; according to Sanquin, the national blood operator of the Netherlands, if policies in Europe don't change, Europe's dependence on the United States will grow to 90 percent in just five years' time.
The same is true for the Anglosphere. New Zealand is the least dependent on paid plasma at a bit more than one-tenth of their need. Australia is now at more than half. The United Kingdom stopped collecting plasma for plasma therapies as a consequence of variant Creutzfeldt-Jakob Disease (and the theoretical threat of it being transmitted through a plasma therapy product), so they are 100 percent dependent on paid plasma. My home country of Canada is now more than 80 percent reliant on American paid plasma. Those proportions are growing in all of these countries, except for the U.K. where there's no possibility to go higher than "all of it."
What gives? Why are the governments and policy makers of Australia, France, Italy, the Netherlands, parts of Canada, and so many other places choosing to pay Americans for their plasma, rather than just permitting their own citizens to be paid for it?
Here's What Gives
Unlike blood, which takes approximately 30 minutes to donate and is possible to do once every 56 days, donating plasma takes about two hours, with 40 minutes of "needle-in" time, and we need people to do it much more often—three times a month or more. Asking people to dedicate two hours a week for an entire year in exchange for cookies and milk is demonstrably asking too much.
Some theorize we just haven't asked in the right way. A recurring fantasy is that all we need is a smashing marketing campaign to make people aware of the need, which will lead to lines out the door to donate at unpaid plasma centers. But this hasn't happened anywhere—not in Spain, Switzerland, Australia, or France.
Millions have been spent on TV, radio, and newspaper advertisements, but those millions would have been better spent if they'd been used to pay donors. The Czech Republic legalized compensation for plasma donations in 2008. Within three years, plasma donations increased sevenfold, with no impact on unpaid blood donations, making the Czech Republic self-sufficient in plasma therapies.
Donor recruitment and retention, staffing, plus marketing costs, combine to make the collection of unpaid plasma two to four times more expensive than just giving money to the donors. A liter of commercial plasma costs about $150–200 on the international market (it averaged $155 in 2017). In comparison, domestic collections in Canada are predicted to cost about $412 per liter by 2024 (if provincial and territorial governments agree to fork over CA$855 million for Canadian Blood Services to open and operate 40 unpaid plasma centers over seven years). According to economist Robert Slonim at the University of Sydney, Australia could save AU$200 million every year if they just stopped collecting unpaid plasma altogether and followed the U.K. model of being 100 percent dependent on America's paid plasma.
#PlasmaGate
The widespread, mistaken belief that unpaid donation is best is a product of the WHO's contrary-to-the-evidence recommendations. In a blunder bigger than their incorrect guidance on masks early in the coronavirus pandemic, the WHO has been pushing for non-remunerated plasma collections for more than four decades, despite this advice being outdated for at least 25 years. That was the last time a plasma therapy was responsible for the transmission of an infection or virus.
The specific recommendation is called "Voluntary Non-Remunerated Blood Donations" (VNRBD). It is intended to capture both blood and plasma donations for purposes of transfusion, as well as plasma donations intended for further manufacture into plasma-derived medicinal products, or plasma therapies. It was motivated by the concern that payment attracted people from lower socioeconomic rungs of the economic ladder who are more likely to be carriers of HIV, hepatitis C, and other transfusion-transmissible infections.
But those concerns no longer apply, partly due to significant improvements in testing technology since the 1970s when the WHO first recommended not paying blood and plasma donors. This improvement in testing happens to form the backbone of arguments among advocates of eliminating restrictions on blood and plasma donation by gay men, which currently require three months of celibacy per the Food and Drug Administration's revised guidance issued this April. But improvements in testing alone are not the reason why plasma for plasma therapies should be considered categorically different from blood and plasma used for transfusions; it is manufacturers' ability to use virus removal and inactivation techniques that marks the stark difference.
In the 1980s, we discovered that heat treatment was effective against HIV. Much like how washing your hands with soap destroys the coronavirus, use of solvents and detergents are effective against lipid-enveloped viruses, including hepatitis C and HIV. Nanofiltration ensures that only molecules of a certain size—the proteins we want—get through, preventing larger molecules from passing into the plasma pool. Most American paid plasma collection centers are also International Quality Plasma Program (IQPP) certified. This voluntary standard, issued by the Plasma Protein Therapeutics Association, involves additional safety steps including the requirement that any donor's first donation be placed on hold, only to be released with the second donation from the same donor. This holding step gives us an opportunity to test the same plasma twice, avoiding the rare possibility of a virus being within the window period where it cannot be detected. This hold means that if you give plasma once and don't go back, your plasma will be discarded.
The result is that, from a scientific and medical perspective, there is no difference between a plasma therapy made from paid plasma and one made from unpaid plasma. There is no additional safety benefit, and the therapy is just as effective.
The WHO knows all about this and so do the national blood operators and regulatory bodies in every country that imports plasma therapies from the United States. They are not concerned about the safety of plasma therapies, but they may be concerned about something else—their employees.
Unionists and Bloodletters
In many countries, the national blood operator is taxpayer-funded. It is common for these organizations to be required to hire employees represented by public sector unions. When that is so, a commercial plasma collector represents a threat to the labor monopoly and a much cheaper alternative to increasing government budget allocations to them for inefficient plasma collections. This suggests a Bootleggers and Baptists-style explanation for prohibition.
Canada gives us a good case study of who the opponents of domestic paid plasma are and what their arguments would be. The only groups that opposed commercial paid plasma were public sector unions and allied activist groups that were either founded or funded by those public sector unions. Those unions succeeded in getting a law passed—called the Voluntary Blood Donations Act—in the provinces of Ontario (2014), British Columbia (2018), and even in conservative Alberta (2017).
They argued that paid plasma is unsafe. It's as though they thought no one would find out that Canada was already relying on American paid plasma for more than 70 percent of its supply of plasma therapies back in 2014. But uncomfortable questions like why they don't oppose imports of paid plasma were never seriously raised.
They also trotted out the bogeyman of American Big Plasma. They don't differentiate Spain-based Grifols from Australia-based CSL Plasma from Japan-based Takeda, the three largest plasma companies, each with hundreds of plasma collection centers in the U.S. They don't appear to understand that these companies compete against each other, nor that Canadian Plasma Resources and Prometic Plasma Resources—the two Canada-based plasma collection companies—represent a competitive threat to American Big Plasma.
Invariably, they also provide the shopworn argument that paid plasma is exploitative without acknowledging that $30-50 for roughly 90 minutes is a fair deal, representing a split of about 30 percent of the revenue to the donor, and 0-5 percent in profit for the company. It is also hard to see how prohibiting paid plasma improves the lot of the poor who sell their plasma, or would if the option were available.
Patient groups representing those whose very lives depend on plasma therapies made from U.S. paid plasma were and are all opposed to these bans. But no one—not the media, not the unions, and not the politicians—seemed to pay that fact any mind.
Bloody Well Pay Them
There is a drop in supply. There may also be a shock to demand as well if the new COVID-19 antibody therapies deliver on their promise.
A lot depends on what happens to plasma donations in the U.S. over the next two or three months. If they recover enough, all will be well, and patients will have uninterrupted access to their treatments. But this will not be the first time patients have had to deal with concerns about supply. In the summer and fall of 2019, a shortage of immune globulin affected not just patients in other countries but patients in the U.S. as well.
"There's something inherently unfair about a system where patients go through a shortage when they live in the same country that provides the vast majority of the global plasma supply," said John Boyle, president of the Immune Deficiencies Foundation. It's so much worse than just "unfair."
Paid plasma collections are the only way that we know of to ensure a safe, secure, and sufficient supply of plasma to meet the growing global need. They are more than twice as effective at less than half the price. Instead of worrying about jobs or imagining that the perfect marketing campaign can bring out volunteers, we should worry about patients instead and just bloody well pay donors already.
